 If ∆S is negative, then the negative signs (from the subtraction and the sign of ∆S) will cancel out, and so as T∆S gets bigger, ∆G will get more positive. T is always positive, so if ∆S is positive then a bigger T∆S will make ∆G more negative (since we subtract T∆S). The chemical equation is: 2 NO 2 (g) -> N 2 O 4 (g) For this reaction, E is -54.72 kJ. As T increases, the T∆S component gets bigger. ∆H is still positive and ∆S is still whatever sign you figured out above. Since ∆H and ∆S don't change significantly with temperature (given in the question), we can assume that they keep the same signs and values: i.e. Chemistry Chemistry questions and answers A) For the reaction 4HCl (g) + O2 (g)->2H2O (g) + 2Cl2 (g) Delta H -114.4 kJ and Delta S -128.9 J/K The equilibrium constant for this reaction at 306. 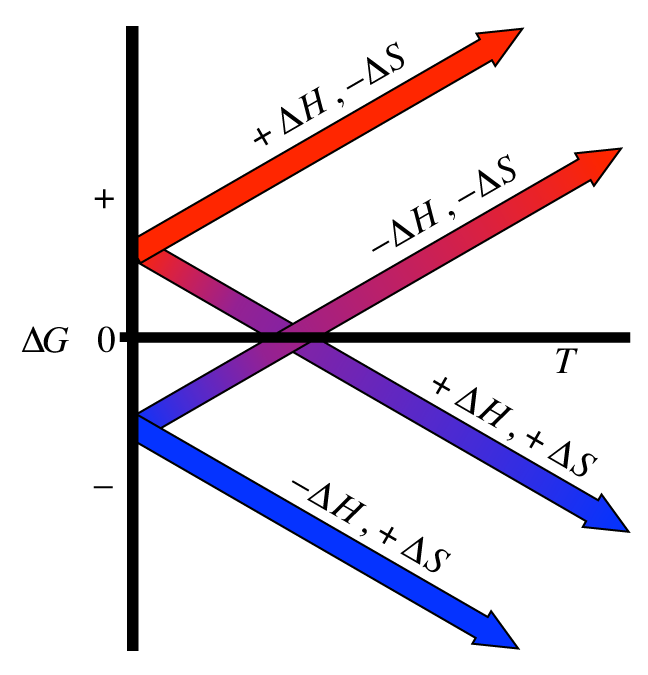
Assume that delta H and delta S are constant. Enter above or below in the first line and enter the temperature in the second line. If ∆G is negative (from the question), is the reaction spontaneous or non-spontaneous?Ģ) Let's use ∆G = ∆H - T∆S again. Question For the reaction 2H2(g) + O2(g)2H2O (g) delta H -484 kJ and delta S -88.9 J/K delta G would be negative at temperatures (above, below) K. From these values, we can know for certain whether ∆S is positive or negative (hint: remember that we are subtracting ∆G!).ġ) Knowing the sign of ∆G is enough to say whether the reaction is spontaneous or not under these conditions. Temperature is always positive (in Kelvin). Calculate Delta H for the reaction 2C(s) + H2(g) arrow C2H2(g) using the following equations. We know (from the question) that ∆G is negative and that ∆H is positive. Thus although the free energy always falls when a gas expands or a chemical reaction takes place spontaneously, there need be no compensating increase in energy anywhere else.This looks like a homework question, so I'll give you some hints to get you on the riht path rather than answering directly.ģ) We know that ∆G = ∆H - T∆S. asked 05/20/21 Calculate the delta H for S + O2 SO2 Given the following data: S + 3/2 O2 SO3 delta H-395.2kJ 2SO2 + O2 2SO3 delta H -198.2kJ Follow 2 Add comment Report 2 Answers By Expert Tutors Best Newest Oldest Paolo S. delta H H (product) - H reactant +ve value. (endothermic) Hence H (product) > H reactant. Reaction is decomposition reaction and needs energy to break H2O. S (product) > S (reactant) deta S S (product) - S (reactant) +ve. It means randomness is increasing from reactant to product. G < 0, the reaction is spontaneous in the forward direction, thermodynamically favourable. reactant has only 2 moles and products have three moles. Calculate the equilibrium constant for the reaction. 
P4O10(s) + 6H2O(l) to 4H3PO4(s) The Delta G of the reaction is -7.050 kJ mol-1. 2Mg(s) + O2(g) 2MgO(s) Calculate Delta H, Delta S, and Delta G for the following reaction at 25 degC. You will have to look up the thermodynamic information. For a given reaction if: G > 0, the reaction is nonspontaneous in the forward direction, not thermodynamically favourable. Calculate delta G for the reaction at 25oC. Free Energy is not energy: A much more serious difficulty with the Gibbs function, particularly in the context of chemistry, is that although G has the units of energy (joules, or in its intensive form, J mol –1), it lacks one of the most important attributes of energy in that it is not conserved. Look at it from the point of view of Gibbs Free Energy, G H T S.  
This is most commonly in the form of electrical work (moving electric charge through a potential difference), but other forms of work (osmotic work, increase in surface area) are also possible. Learning Objectives To get an overview of Gibbs energy and its general uses in chemistry. By "useful", we mean work other than that which is associated with the expansion of the system. The “free” part of the older name reflects the steam-engine origins of thermodynamics with its interest in converting heat into work: ΔG is the maximum amount of energy which can be “freed” from the system to perform useful work. Free Energy is not necessarily "free": The appellation “free energy” for G has led to so much confusion that many scientists now refer to it simply as the Gibbs energy. Click here to get an answer to your question If S + O2 SO2 H - 398.2 kJ : SO2 + 12O2 SO3 H - 98.7 kJ SO3 + H2O H2SO4 H - 130.2. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
